INDICATIONS AND USAGE
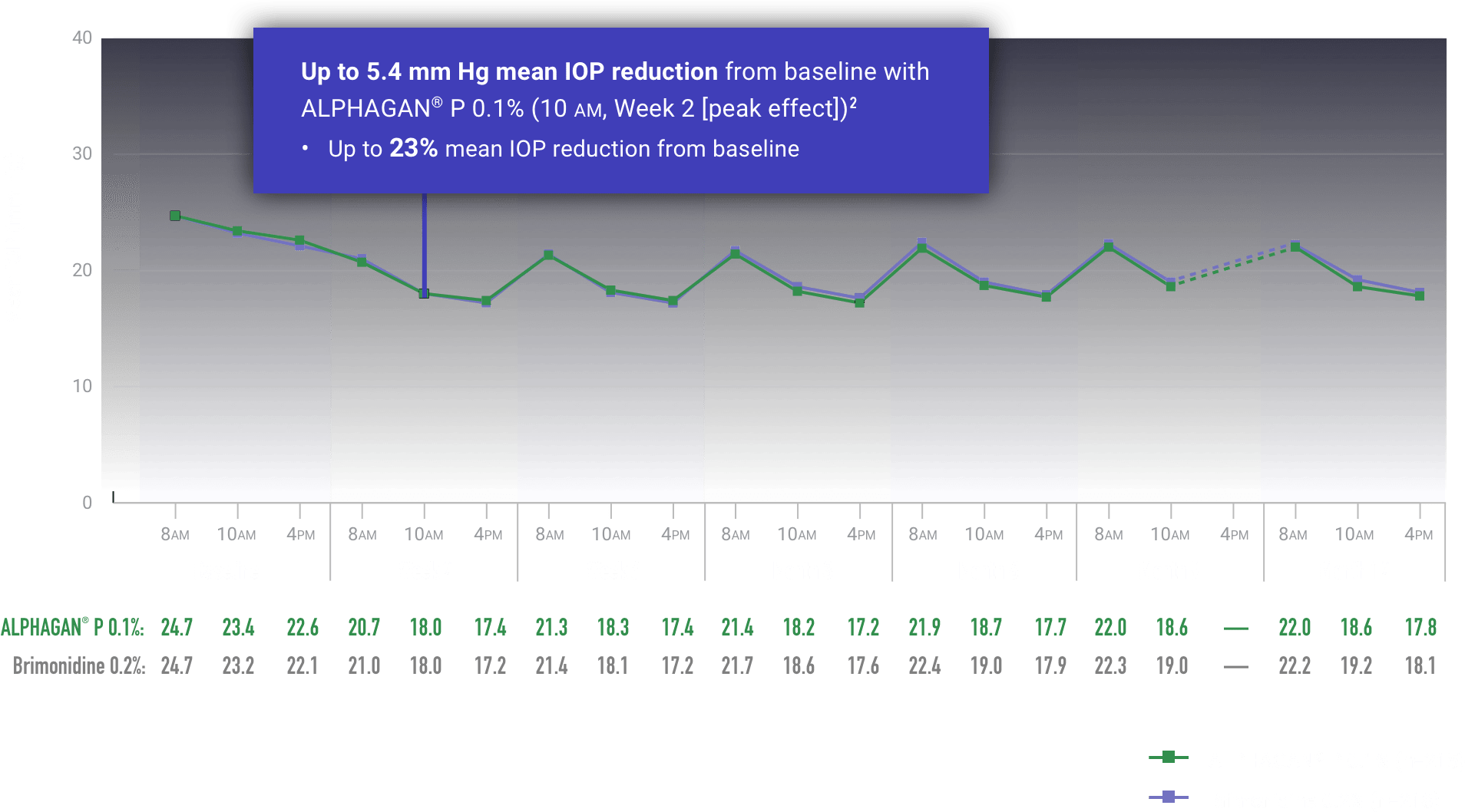
ALPHAGAN® P (brimonidine tartrate ophthalmic solution) 0.1% or 0.15% is indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Neonates and Infants (pediatric patients younger than 2 years old): ALPHAGAN® P is contraindicated in neonates and infants (pediatric patients younger than 2 years old).
Hypersensitivity Reactions: ALPHAGAN® P is contraindicated in patients who have exhibited a hypersensitivity reaction to any component of this medication in the past.
WARNINGS AND PRECAUTIONS
Potentiation of Vascular Insufficiency: ALPHAGAN® P may potentiate syndromes associated with vascular insufficiency. ALPHAGAN® P should be used with caution in patients with depression, cerebral or coronary insufficiency, Raynaud's phenomenon, orthostatic hypotension, or thromboangiitis obliterans.
Severe Cardiovascular Disease: Although brimonidine tartrate ophthalmic solution had minimal effect on the blood pressure of patients in clinical studies, caution should be exercised in treating patients with severe cardiovascular disease.
Contamination of Topical Ophthalmic Products After Use: There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface. Do not touch the tip of the dispensing container to the eye or surrounding structures. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions.
DRUG INTERACTIONS
Antihypertensives/Cardiac Glycosides: ALPHAGAN® P may reduce blood pressure. Use caution in patients on antihypertensives and/or cardiac glycosides.
CNS Depressants: Although specific drug interaction studies have not been conducted with ALPHAGAN® P, the possibility of an additive or potentiating effect with CNS depressants (alcohol, barbiturates, opiates, sedatives, or anesthetics) should be considered.
Tricyclic Antidepressants: Tricyclic antidepressants have been reported to blunt the hypotensive effect of systemic clonidine. It is not known whether the concurrent use of these agents with ALPHAGAN® P in humans can lead to resulting interference with the IOP-lowering effect. Use caution in patients on tricyclic antidepressants, which can affect the metabolism and uptake of circulating amines.
Monoamine Oxidase Inhibitors: Monoamine oxidase (MAO) inhibitors may theoretically interfere with the metabolism of brimonidine and potentially result in an increased systemic side effect such as hypotension. Use caution in patients on MAO inhibitors, which can affect the metabolism and uptake of circulating amines.
ADVERSE REACTIONS
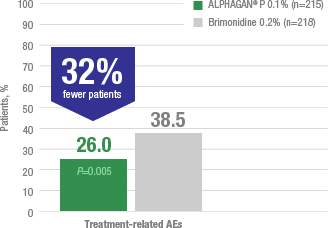
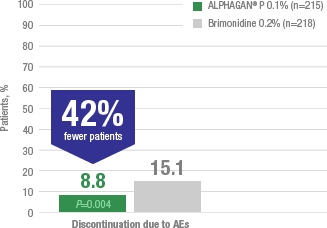
Adverse reactions occurring in approximately 10% to 20% of the subjects receiving brimonidine ophthalmic solution (0.1% to 0.2%) included: allergic conjunctivitis, conjunctival hyperemia, and eye pruritus. Adverse reactions occurring in approximately 5% to 9% included: burning sensation, conjunctival folliculosis, hypertension, ocular allergic reaction, oral dryness, and visual disturbance.
Please see accompanying full Prescribing Information or visit
https://www.rxabbvie.com/pdf/
alphagan_pi.pdf